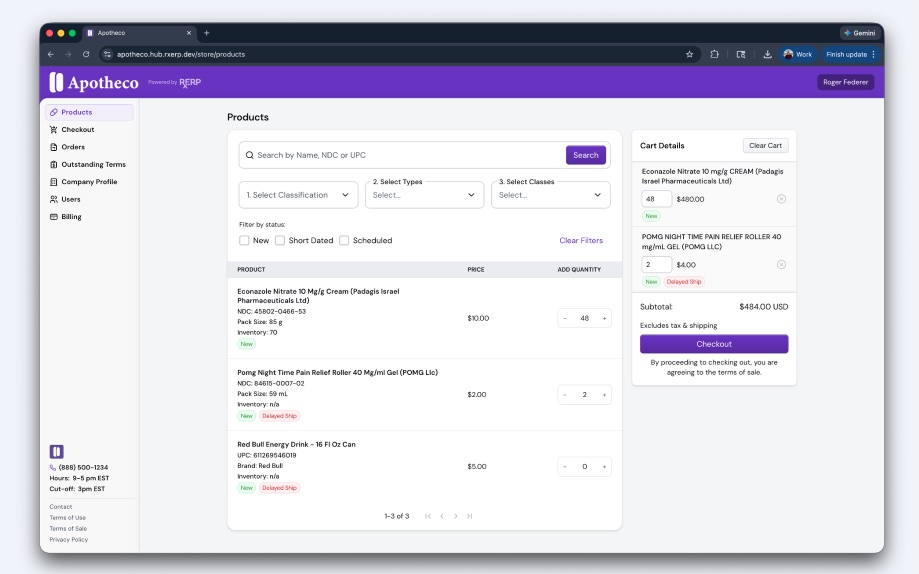
Steve Madsen hit his breaking point when compliance became unmanageable. Running a pharma wholesale distributor meant juggling Drug Supply Chain Security Act (DSCSA) serialization across eight disconnected systems, each adding operational and regulatory risk with every transaction.
The problem was infrastructure. RxERP emerged from that pain: a serialized ERP built to unify data, workflows and compliance into a single platform. With unit-level audit logging and a hub that acts as the source of truth, RxERP aims to move pharma from reactive reconciliation to real-time, provable trust across the entire supply chain.
Madsen shares his insights in this Q&A on how they overcame these challenges and the implications for other vendors not just in pharma, but in other industries, as well.
Question: RxERP was built out of your own experience running a pharmaceutical wholesale distributor. Can you describe the specific breaking point that made you realize the industry needed a fundamentally different approach to serialized ERP?
Steve Madsen: The breaking point came when we realized compliance wasn’t just inefficient, it was unmanageable at scale. Running a wholesale distributor, we were juggling DSCSA serialization, fragmented data and manual reconciliation across multiple systems that didn’t talk to each other. In our case, it meant operating across eight separate systems just to try and stay compliant. Every transaction carried operational and regulatory risk. It became clear the industry wasn’t lacking effort; it lacked infrastructure. RxERP was built to unify it all into one platform.
Q: Unit-level audit logging is a centerpiece of this release. How does having a serialized audit trail built into the ERP by default change how pharmaceutical companies approach investigations, recalls or regulatory scrutiny?
SM: Unit-level audit logging shifts pharma from reactive investigations to real-time traceability. Instead of reconstructing events across fragmented systems, every serialized unit carries a complete, immutable history within the ERP. This dramatically reduces recall scope, accelerates root-cause analysis, and strengthens audit readiness. Under regulatory scrutiny, companies move from defending data integrity to demonstrating it instantly, with precision down to the unit level reducing risk, cost and operational disruption.
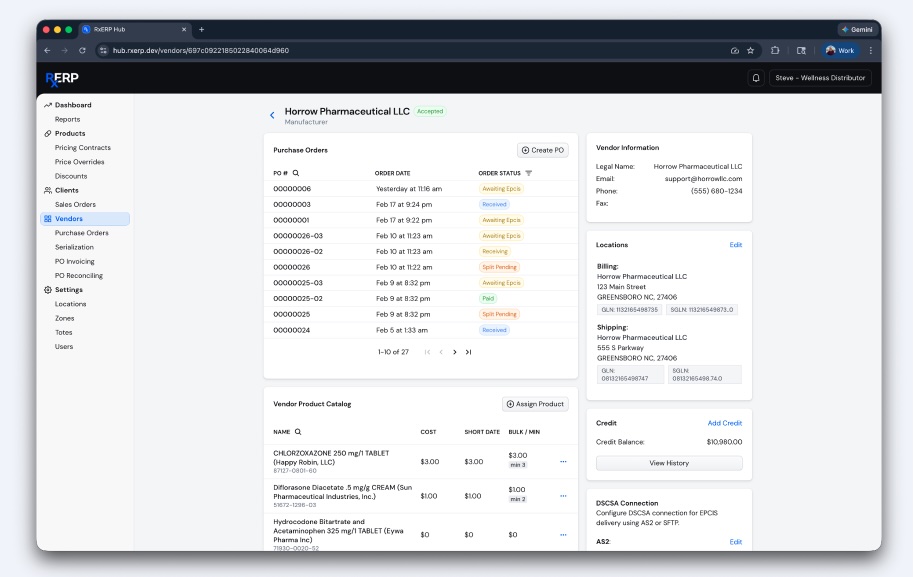
Q: You said the Hub “creates the sources of truth the pharma supply chain has been missing.” What are the consequences when that source of truth is absent and what goes wrong downstream for manufacturers, distributors and pharmacies?
SM: When a central source of truth is absent, the supply chain fragments. Manufacturers lose visibility into product movement and risk compliance gaps. Distributors face reconciliation errors, duplicate or mismatched data, and increased audit exposure. Pharmacies operate with inconsistent inventory, leading to dispensing errors or delays. Downstream, this results in failed traceability, broader recalls, higher operational costs, and regulatory risk. Ultimately, the system becomes reactive, forcing every participant to validate data instead of trusting it.
Q: What future automation and AI use cases become possible once pharma organizations can reliably trust RxERP’s unit-level transaction history and proof layer?
SM: Once the data layer is trusted at the unit level, automation moves from rules-based to autonomous. AI can predict shortages, flag anomalies before claims submission and automate recalls with exact precision. Smart contracts can trigger payments or compliance actions in real time. Fraud detection becomes proactive, not forensic. Inventory, financing, and regulatory workflows can self-orchestrate because the underlying data no longer needs validation, unlocking true end-to-end automation across the pharma supply chain.
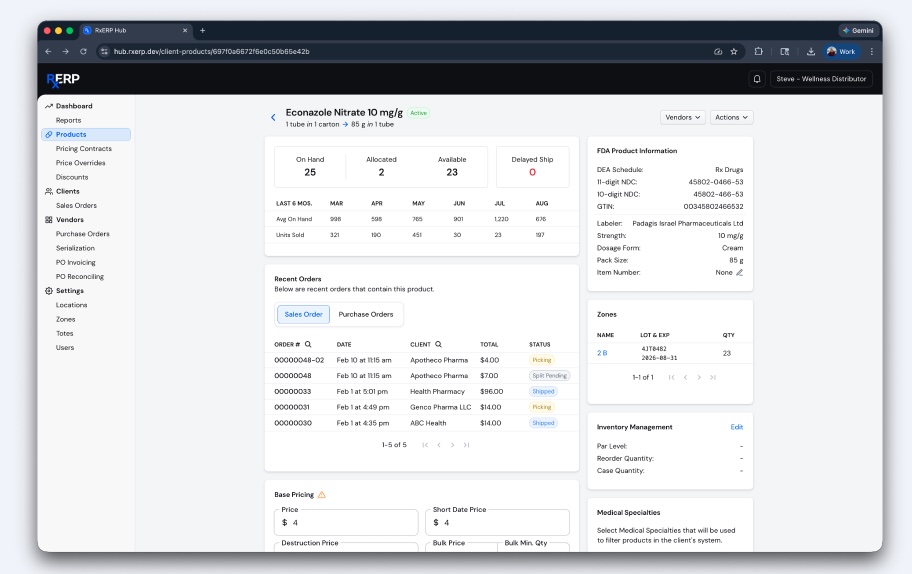
Q: In practice, how does RxERP help pharma organizations protect patients and combat counterfeits while still streamlining logistics and inventory operations?
SM: RxERP embeds serialization, verification and auditability directly into daily workflows, so every unit is tracked, verified, and reconciled in real time. This prevents counterfeit or diverted products from entering the supply chain while creating immediate traceability to the source. At the same time, it replaces manual checks and fragmented systems, streamlining inventory management, order processing and compliance. The result is stronger patient safety with faster, more efficient logistics and significantly reduced operational friction.
Q: How do you see RxERP Hub changing audit readiness expectations for pharmaceutical manufacturer, distributors and 3PLs?
SM: RxERP Hub shifts audit readiness from periodic preparation to continuous compliance. Instead of assembling data for audits, manufacturers, distributors and 3PLs operate with always-on validation, complete traceability, and immutable records at the unit level. Auditors can access standardized, real-time data rather than fragmented reports. This raises expectations across the industry moving from reactive documentation to proactive, system-enforced compliance that reduces audit risk, accelerates inspections and builds immediate trust with regulators.
Q: Looking ahead, how might RxERP’s serialized source-of-truth model influence ERP roadmaps for other highly regulated, safety-critical industries beyond pharma?
SM: RxERP’s serialized source-of-truth model will likely redefine ERP expectations across all safety-critical industries. Sectors like medical devices, aerospace, food, and defense will move toward unit-level traceability, embedded compliance, and immutable audit layers as standard infrastructure. Instead of systems of record, ERPs become systems of proof where every transaction is verified, traceable and trusted in real time. This enables automated recalls, predictive risk detection, and regulator-grade transparency by default. Over time, the market will demand ERPs that don’t just manage operations, but enforce integrity, accountability, and safety at the most granular level possible.